[ad_1]
Antitumor responses mediated by T helper 17 cells can be attenuated by intradermally injected hollow nanoparticles displaying the polysaccharide mannan.
In the late nineteenth century, William Colley, a surgeon at the New York Memorial Hospital, observed a spontaneous tumor remission in a sarcoma patient who had concurrent Streptococcus Infection. Motivated by this observation, Coli prepared a combination of bacteria streptococcus pyogenes And Serratia marcescensand used it as an anticancer therapy in patients1, Although the method was never tested in large randomized trials, and septic complications sometimes overshadowed the antitumor activity, substantial evidence had accumulated for the effects of the so-called E. coli toxins, and by the 1960s, cancer The therapy was systematically used to treat patients with. Today, interest remains high in the use of comparable approaches to elicit antitumor immune responses. coli’s pioneering work was an important indication that activation of the immune system has a role in eradicating cancer. coli toxins were unknown at the time, it is now clear that immune responses are triggered by the activation of pattern-recognition receptors (PRRs) by conserved bacterial motifs known as pathogen-associated molecular patterns (PAMPs). Huh. By taking advantage of nanotechnology, materials can now be designed to display PAMPs to induce an immune response without the risk of severe sepsis. This is exemplified by a study by James Moon and colleagues2published in Nature Biomedical EngineeringReporting the design and development of nanoparticles incorporating structures of the fungal derived polysaccharide mannan Saccharomyces cerevisiae, By engaging the well-known PRRs dectin-2 and toll-like receptor 4 (TLR4) on dendritic cells, the nanoparticles decorated with mannan motifs induced the activation of responses from the innate and adaptive immune system by acting as PAMPs and directly Cause not created. Poisoning.
Fungal Mannan T Helper 17 (Th17) Reactions3, Moon and co-authors now show that mannan-exhibiting nanoparticles strongly induce Th17-mediated lymphocyte differentiation, reducing the balance between Th17 and regulator T (Treggae) activation of cellular immune responses and towards cells away from the inhibitory effect of Treggae cells. In particular, the ability of the nanoparticles to activate both TH17 and innate immune cells led to intratumoral stimulation of CD8 cells and natural killer cells and potent antitumoral effects, as the authors demonstrate in mice treated intratumorally with nanoparticles. The authors also show in mouse models of colon carcinoma and melanoma that the antitumoral effect of the nanoparticles was further potentiated by combining the nanoparticles with an agonistic antibody to the OX40 co-stimulatory receptor.
Tumor infiltration by different populations of immune cells is a hallmark of cancer. Depending on their type and function, immune cells in tumors differentially affect disease progression.4and their function is affected by the tumor microenvironment5,6, Indeed, cancer leads to an increase in myelopoiesis, especially in the production of monocytes, macrophages and neutrophils, and these cells can accumulate in the periphery of the tumor and shift their phenotype to promote tumor growth.6,7, Such immunosuppressive myeloid cells are also a major obstacle to the beneficial effects of checkpoint inhibitors on CD8 activation and tumor eradication.8, In addition, the CD4 lymphocyte population (Th1, TH2 more tH17) release endogenous mediators (such as cytokines) that exert antitumoral immune effects. role of T.H1 CD4 lymphocytes are closely linked to the immunosurvival program, yet little is known about the role of TH17 On lymphocytes in cancer and their potential as immunotherapeutic targets. It has recently been shown that adoptive tH17 cells reduce tumor growth and tumor-associated interleukin-17 (IL-17) is related to survival of cancer patients9,10, But how to induce such antitumor effect in vivo was not clear.
Moon and co-authors’ findings bring new insights on several levels. At the pathophysiological level, they validate the importance of th17 responses involving the induction of antitumoral mechanisms through activation of CD8 and natural killer cells (Fig. 1); This opens the door to future immunotherapeutic approaches. The authors also report a role for dectin-2 and TLR4 in this process; This may facilitate the development of regulatory therapies acting at the level of these receptors, which can be used either through immune suppression (via stimulatory agents), or in conditions characterized by inflammation and autoimmunity (via inhibitory approaches). can be done. At a technical level, the authors show the possibility of engineering nanoparticles containing complex microbial glycans (in their case, Saccharomyces mannan), and that the nanoparticles displayed significant antitumoral effects. Other fungal glycans, notably beta-glucans, have been shown to exert potent antitumoral effects in experimental models of cancer, but these effects are said to arise from the induction of trained innate immunity.1 1, Induction of innate immune activation by mannan nanoparticles may well lead to long-term trained-immune induction (besides T).h17 Discrimination); Whether this is the case will need to be investigated in follow-up studies. and, at the therapeutic level, the authors show strong synergy of Th17 Differentiation induced by mannan-decorated nanoparticles and OX40 activation by agonistic antibodies. This supports the notion that, in general, combination cancer immunotherapies are likely to be much more effective than monotherapies.
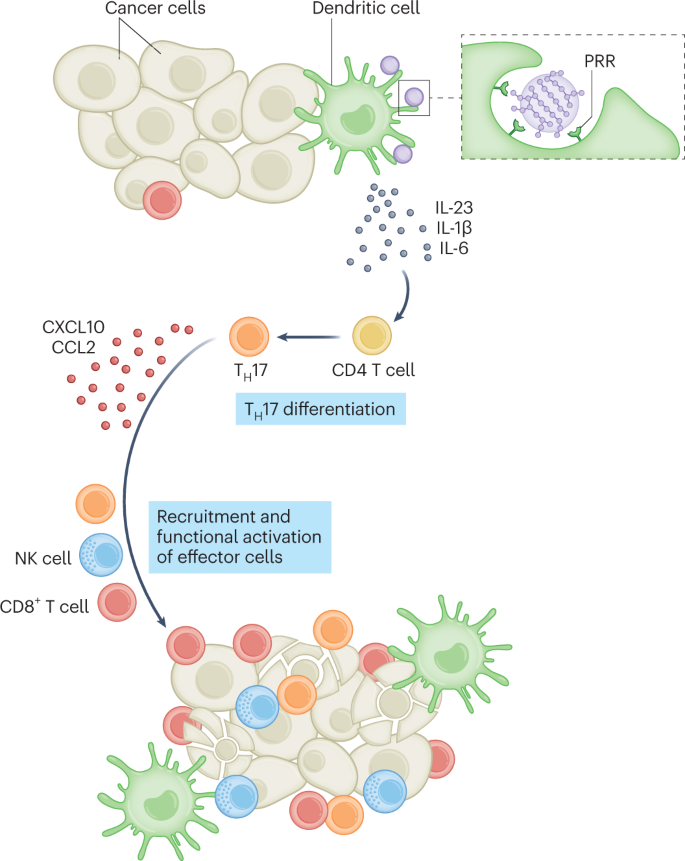
Nanoparticles displaying the fungal polysaccharide mannan intradermally delivered PRR on dendritic cells inducing Th17-mediated lymphocyte differentiation through the interleukins IL-23, IL-1β and IL-6. TeaH17 cells favor the intratumoral recruitment of CD8, T cells and natural killer (NK) cells by releasing pro-inflammatory chemicals (such as CXCL10 and CCL2). Figure adapted with permission from ref. 2, Springer Nature Limited
Mannans are complex structures that interact with multiple PRRs on various innate and adaptive immune cells. The relevance of any additional potential mechanisms through which the mannan-decorated nanoparticles exert their effects, including effects at the level of the innate immune system such as trained immunity (which may also induce antitumor responses)12), further study is needed. Furthermore, there is a need to distinguish between the immune stimulatory and antitumor effects of the nanoparticles themselves. Other nanoparticle systems, such as lipid nanoparticles used in COVID-19 vaccines13, Can get strong anti-inflammatory effect. Therefore, any inflammation-mediated adverse effects thus need to be investigated. The combination of mannan-decorated nanoparticles and anti-OX40 agonistic antibody seems translationally promising, and it would therefore be worth optimizing the nanoparticles for intravenous administration. Furthermore, it will be important to study whether the nanoparticles can also enhance the effects of antibodies against immune checkpoints (such as programmed cell death receptor 1 or cytotoxic T-lymphocyte-associated protein 4), as well as the effects of others. Types of immunotherapy. Nanoparticles containing PAMPS that activate the PRR could lead to new immunotherapies.
[ad_2]
Source link

